Science unlocked: publication picks from June 2025
In this monthly series, we share a selection of recent publications in which Oxford Nanopore sequencing was used to unlock novel insights. Spanning from direct RNA transcriptomics, to complex diseases, to single cell research, these studies showcase the advances in scientific research made possible by Oxford Nanopore sequencing.
Featured in this edition:
1. Broadening the global picture of ataxia
2. The rapid RNA modifications behind type 2 diabetes
3. Bird flu in retail dairy products
4. An RNA atlas of the ageing mouse brain
Human genetics
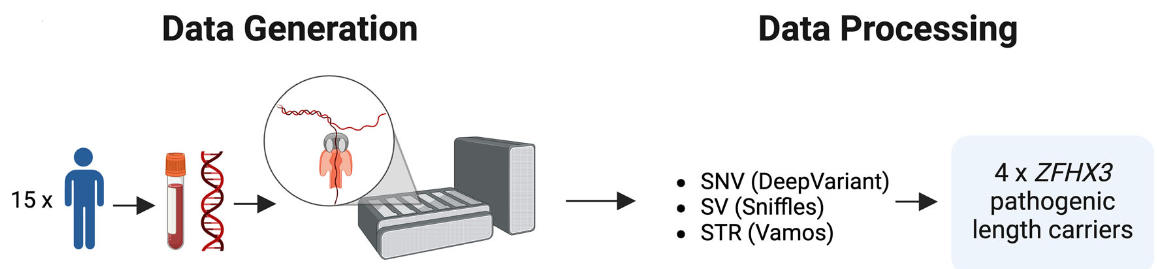
1. Identification of GGC repeat expansions in ZFHX3 among Chilean movement disorder patients (Movement Disorders)
Saffie-Awad et al. identified the first ZFHX3 GGC repeat expansions associated with spinocerebellar ataxia type 4 (SCA4) in Latin America. Their findings link longer repeats to earlier onset, demonstrate repeat instability and epigenetic changes, and illustrate the potential of Oxford Nanopore sequencing for future use in hereditary ataxia research and screening.
Key points:
Four Chilean patients were found to have ZFHX3 GGC repeat expansions (47–55 units), the first such cases outside Northern Europe
All showed classic SCA4 symptoms, and one case also involved parkinsonism, broadening the known clinical spectrum
Longer expansions were associated with more severe disease and earlier onset
One family exhibited intergenerational repeat expansion from 49 to 55 units, confirming repeat instability
Allele-specific hypermethylation was observed, suggesting an epigenetic role in disease pathogenesis
No repeats ≥42 units were found among 438 diverse controls, reinforcing the threshold for pathogenicity
Figure: schematic overview of the study design. Blood samples were taken from 15 individuals with suspected hereditary movement disorders. The extracted DNA was sequenced on a PromethION device. Variant calling included single nucleotide variants (SNV), structural variants (SV), and short tandem repeats (STR). The researchers identified pathogenic ZFHX3 GGC expansions in four individuals. Figure redistributed from Saffie-Awad et al. 2025 under Creative Commons Attribution License CC BY 4.0.
Watch Paula’s talk at London Calling 2025, or catch the highlights in the vox pop below
Mulroney, Taylor, and Lee et al. used Oxford Nanopore direct RNA sequencing to investigate how glucose levels affect RNA modifications in human pancreatic beta cells, uncovering thousands of glucose-responsive changes. The study reveals that these modifications, especially m⁶A, are enriched in type 2 diabetes–related genes and act independently of gene expression, offering new insights into RNA-based regulation in diabetes.
Key points:
The team profiled four RNA modifications: m⁶A, m⁵C, inosine, and pseudouridine
Of the 2.7 million sites detected, 1,697 showed significant changes after glucose stimulation
Most modification changes occurred without changes in gene or transcript expression, indicating distinct regulatory mechanisms
m⁶A dominated among altered sites and was enriched in key diabetes genes
This is thought to be the first report of glucose-responsive changes in m⁵C, inosine, and pseudouridine in beta cells
Oxford Nanopore sequencing enabled simultaneous, isoform-level analysis of all modifications in one assay
Figure: EndoC-BH1 (top) and EndoC-BH3 (bottom) were cultured in low glucose condition (2.8 mM, blue) or high glucose condition (15.0 mM, yellow). RNA was extracted from each culture and used to prepare native direct RNA sequencing libraries (which retain RNA modifications), or in vitro transcribed (IVT) RNA sequencing libraries, where RNA modifications are lost during complementary (c)DNA synthesis. Figure redistributed from Mulroney, Taylor and Lee et al. 2025 under Creative Commons Attribution License CC BY 4.0.
See what insights you could unlock with Oxford Nanopore direct RNA sequencing
Infectious disease
Lail et al. used tiled-amplicon Oxford Nanopore sequencing to detect and characterise H5N1 virus in pasteurised dairy, achieving over 90% genome coverage in viral RNA-positive samples. This method offers a rapid, accessible surveillance solution in regions without on-farm testing, supporting early detection and response to viral spread.
Key points:
Highly pathogenic avian influenza viruses are infecting humans and US dairy cattle, raising public health concerns
Surveillance gaps exist where on-farm testing is unavailable
Oxford Nanopore sequencing provided over 90% genome coverage at greater than 20x depth in 5/13 viral RNA-positive dairy samples
Viral sequences clustered with other cattle outbreak strains but showed no state-level grouping
Their findings suggest either inter-state viral movement or limited sequence variation
A scalable approach, such as Oxford Nanopore sequencing, could enhance genomic surveillance using easily accessible sample types
Early detection from retail products may aid a rapid response and outbreak prevention
Read more on viral amplicon sequencing with Oxford Nanopore technology
Single cell
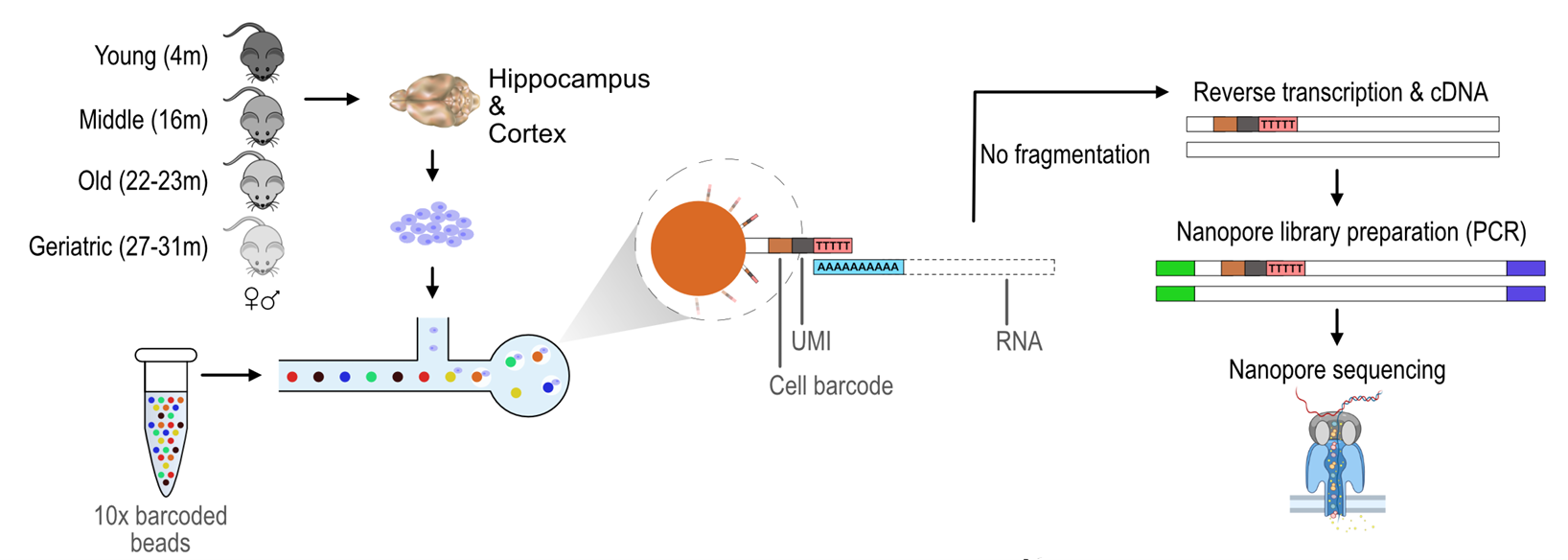
4. Cell-specific RNA isoform remodelling in the ageing mouse brain (bioRxiv)
Using Oxford Nanopore sequencing, Rehman et al. built a full-length RNA isoform atlas in two regions of the mouse brain, revealing thousands of cell-type and age-related transcript changes. This enabled unprecedented insights into ageing biology and neurodegeneration, offering a path to earlier detection and intervention for age-linked brain disorders in the future.
Key points:
The atlas captured transcriptomic changes across 51 cortex and hippocampus samples from male and female mice across four age groups
Rehman et al. identified over 3,700 cell-type-specific and over 4,700 age-associated isoform changes
Immune and oligodendrocyte cells showed the greatest isoform diversity
Ageing was linked to longer, coding isoforms due to shifts in transcription start and end sites
Neuronal isoforms were most strongly downregulated with age, suggesting significant transcriptomic remodelling and potential vulnerability to neurodegeneration
The proportion of senescent immune cells rose sharply with age, particularly in the hippocampus and in females
Oxford Nanopore sequencing enabled the analysis of full-length native RNA at the single-cell level, offering a powerful tool for studying ageing biology and advancing early detection strategies for brain disorders
Figure: schematic of experimental workflow to map the cellular and regional transcriptomic landscape of the ageing mouse brain with Oxford Nanopore sequencing. Hippocampus and cortex samples were taken postmortem from male and female mice from across four age groups. The brain tissue was dissociated into single cells, which were then encapsulated individually in droplets with a barcoded bead and reverse transcriptase. Each RNA transcript was tagged with a unique molecular identifier (UMI). The barcoded cDNA from all the cells was then amplified for Oxford Nanopore sequencing. Figure made available for use by Rehman et al. under a CC0 license.
Check out our workflow overview for single-cell transcriptomics
Inspired? Apply Oxford Nanopore sequencing to your own research questions and you'll never see sequencing the same way again. Explore the nanopore sequencing solution.
Oxford Nanopore Technologies products are not intended for use for health assessment or to diagnose, treat, mitigate, cure, or prevent any disease or condition.
Saffie-Awad, P. et al. Identification of GGC repeat expansions in ZFHX3 among Chilean movement disorder patients. Movement Disorders (2025). DOI: https://doi.org/10.1002/mds.30242
Mulroney, L., Taylor, H.J. and Lee, A. et al. Direct RNA nanopore sequencing reveals rapid RNA modification changes following glucose stimulation of human pancreatic beta-cell lines. bioRxiv 659352 (2025). DOI: https://doi.org/10.1101/2025.06.12.659352
Lail, A.J. et al. Amplicon sequencing of pasteurised retail dairy enables genomic surveillance of H5N1 avian influenza virus in United States cattle. PLOS One (2025). DOI: https://doi.org/10.1371/journal.pone.0325203
Rehman, A. et al. Cell-specific RNA isoform remodelling in the ageing mouse brain. bioRxiv 658133 (2025). DOI: https://doi.org/10.1101/2025.06.05.658133